|
12/30/2023 0 Comments Chemdoodle demo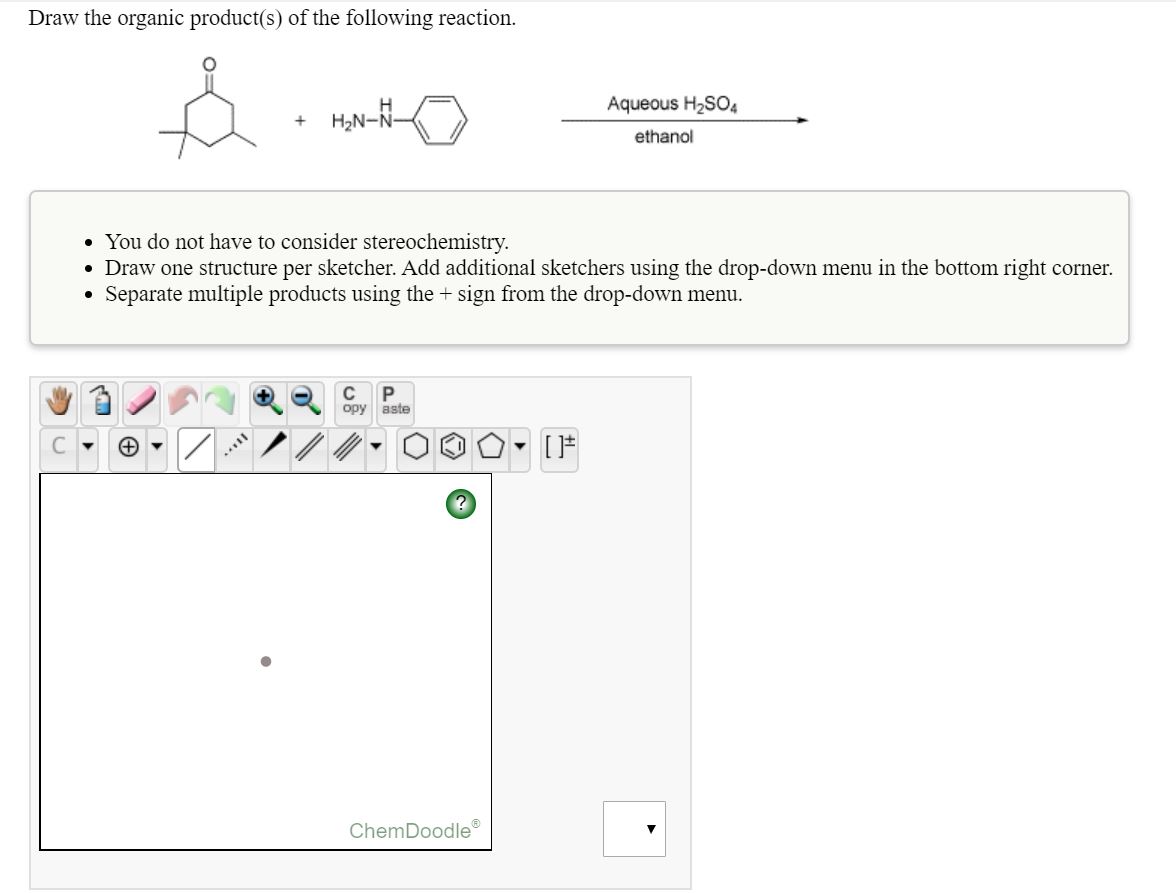
Therefore, canonical SMILES is not a solution, and you need to use ChemDoodle's fignerprinting and isomorphism engine instead. Superstructure, query or maximum common substructure. Canonical string comparisons are also only applicable to exact matching, and not substructure, About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy & Safety How YouTube works Test new features NFL Sunday Ticket Press Copyright. Handle molecule structure comparisons is through graph isomorphism implementations, perhaps after a fingerprint Integrity of your dataset will eventually be dependent on using old and obsolete software. Software implementations continuously change, invalidating previously generated canonical output. One using a canonical SMILES string for a structure would be confident in matchingĪnother structure converted into a SMILES string if the two strings are identical. 
Canonical SMILES algorithms today are unique to the developer and are not cross-compatible.Ĭanonicalization is an attempt to simulate graph theoretical functionality without making use of graph Help to make a smooth process either is generate good graphics, modern tools, reactions, calculators, compatibility with 3d models, cheap in a process and customizable by users requirements. Flexible run on mobile gadgets and web browsers. Protocol, but Daylight Informatics did publish a vague and incomplete CANGEN algorithm for canonicalizing ChemDoodle is an Online drawing tools for chemical industries. Canonical SMILESĬanonicalization is typically used for indexing and exact matching in databases. This demo uses a Century Gothic web safe font for thinner, accentuated sans-serif text that matches the bond strokes in chemical structures. Please give it a try and let us know if you encounter any issues. ChemDoodle Web Components allow the wielder to present publication quality 2D and 3D graphics and animations for chemical structures, reactions and spectra. When you are done drawing, press the Output SMILESīutton, and the generated SMILES string will be displayed in the text area. Multiple structures, or a single reaction. SMILES strings can be generated for drawn structures in the sketcher. ChemDoodle provides aīest-in-class 2D coordinate generation algorithm (including expert stereochemistry diagram support), please see this demo for more information.Ĭoordinates, but this SMILES demo is limited to 2D output. Input, as SMILES strings contain no coordinate information. One of the main requirements for a SMILES parser is to generate coordinates for the SMILES string, ChemDoodle will still assume it is a SMILES string and try to parse it.įor describing errors and warnings in input SMILES string. ChemDoodle Web Components allow the wielder to present publication quality 2D and 3D image and animations for chemo structures, reactions and spectra. Please note, even if you type in a random string of characters that is clearly not a Please give it a try and let us know if you encounter any issues. We provide some complexĮxamples as well, just click on the example to load it. Will be rendered in the ChemDoodle Web Components viewer above. 
Input the SMILES string you want to parse an depict in the text field above, and then
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
