|
12/29/2023 0 Comments Boiling point of liquid oxygen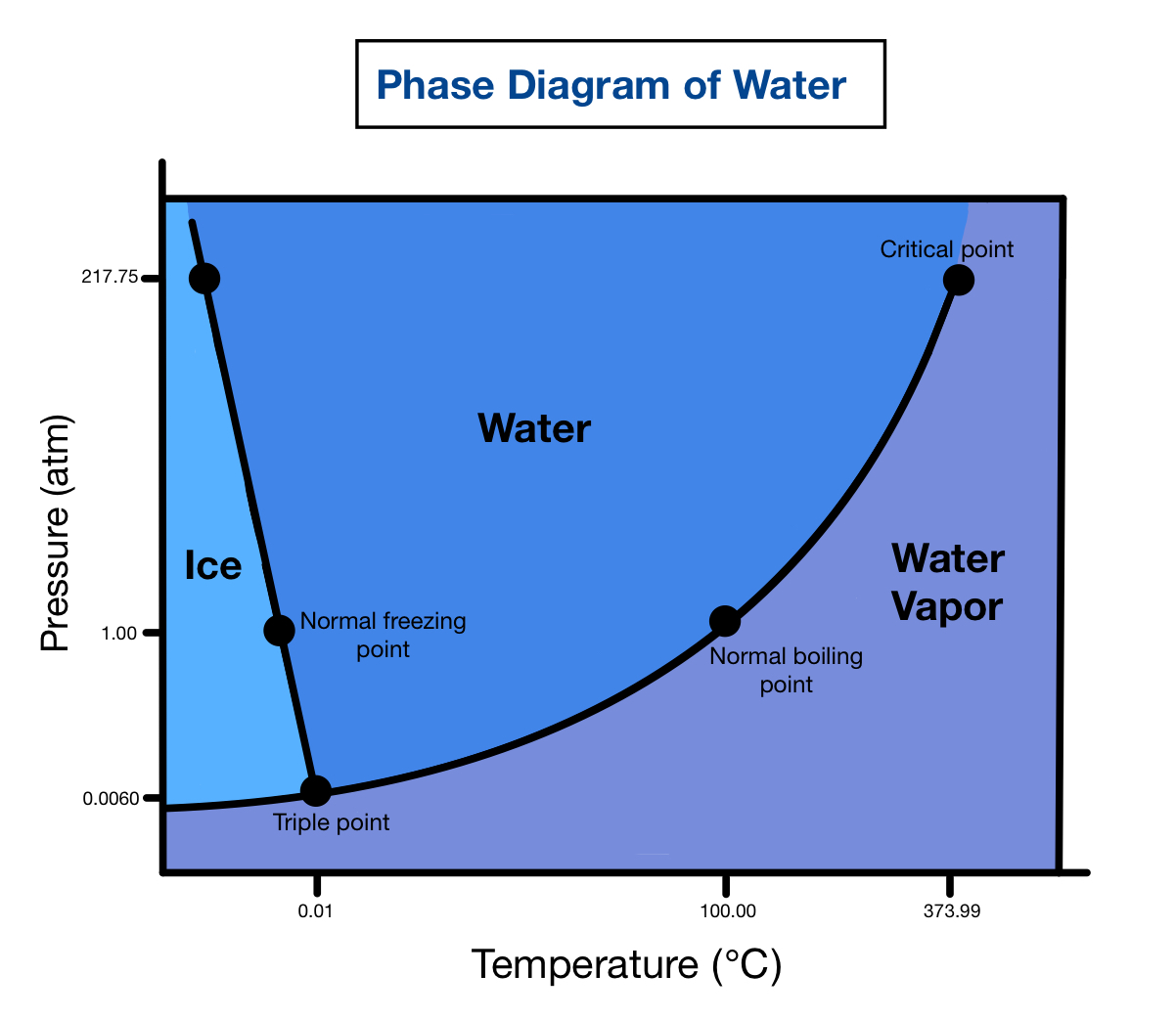
Examples of other hazards that can be present include: In addition to the characteristics shared by all refrigerated and cryogenic liquids, each product has its own unique hazards that should be understood and managed through safe handling practices. Once liquefied, all refrigerated and cryogenic liquids must stay extremely cold or they will return to a gas state in a process known as vaporization.Īll refrigerated and cryogenic liquids have several common properties: Likewise, refrigerated and cryogenic liquids turn into gas above their boiling points, and are liquids at temperatures below their boiling points. Between the boiling point and 32 ☏ (0 ☌), water is a liquid. 
Above this temperature, water vaporizes into a gas (steam). For example, the boiling point for water is 212 ☏ (100 ☌). Cryogenic liquids are defined as having a boiling point below –130 ☏ (–90 ☌). Refrigerated and cryogenic liquids are gases that change to a liquid once they drop below a certain temperature, known as the boiling point, which is different for each product. You should always read and understand the label and the SDS before using any product and should also follow the instructions and safety precautions provided by your product supplier. The container label and safety data sheet (SDS) provide detailed hazard information and handling precautions. The specific gas product being used can pose other hazards in addition to being a refrigerated or cryogenic liquid. This safety poster, provided by the Compressed Gas Association, provides basic safety information for the safe use of refrigerated and cryogenic liquids. In addition to being extremely cold, other hazards include high rates of vaporization, rapid expansion, and fogging. 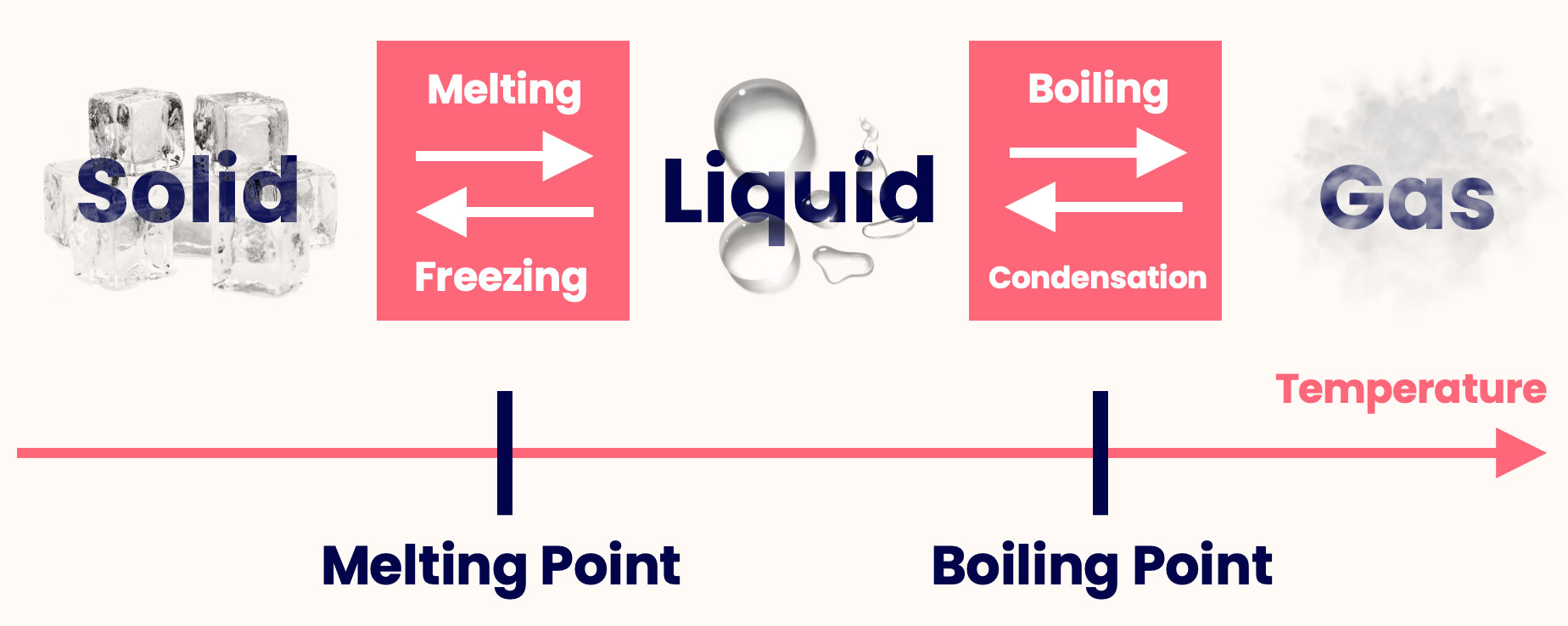
Once liquefied, all refrigerated and cryogenic liquids must stay extremely cold or they will return to a gas state in a process known as vaporization.Īnyone who handles refrigerated or cryogenic liquids should be aware of their unique properties and potential hazards. These liquids are extremely cold cryogenic liquids are defined as having a boiling point below –130 ☏ (–90 ☌). Note: There are some serious health hazards related to the liquified oxygen at concentrations more than 80% for a longer period of time, then it can cause irritation in the lungs, nose stiffness and sore throat.Refrigerated and cryogenic liquids are gases that have become a liquid due to very low temperatures. Vaporizers convert the liquid oxygen into gas and the pressure system maintains the pressure. The construction of a cryogenic tank is similar to an insulated thermos bottle, there are two layers separated from each other with a distance, the outer layer protects the liquid oxygen to be in contact with the surrounding. If we look at the storage system of liquid oxygen in brief, then there is a storage system with a cryogenic tank, vaporizers, pressure control system and piping. The equipment of handling liquid oxygen is also very specific as it is mostly liquified to save the cost of transporting and storing. Cryogenic liquids are liquified gases whose normal boiling point is below $-150^\circ C$, because it is very essential to keep the liquid oxygen protected from the surrounding heats even in winters otherwise, it has its own health hazards. The oxygen can react with all the metals forming their oxides and react very vigorously as a substance which burns in air will readily burn in the presence of oxygen. It Is non-flammable but a very strong oxidizer. The liquid oxygen is pale blue in colour and very cold. Hint: Oxygen is the second largest gas of the atmosphere having 20.8% volume existence.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
